As a result of cold working, the hardness, tensile strength, and electrical resistance increases, while ductility decreases. There is also a large increase in the number of dislocations, and certain planes in the crystal structure are severely distorted. Although most of the energy used to cold work the metal is dissipated in heat, a finite amount of energy (about 1 to 10 percent) is stored in the crystal structure as internal energy associated with the lattice defects created by the deformation. Full annealing is the process by which the distorted cold worked lattice structure is changed back to one which is strain free through the application of heat. Information about annealing and hot working is given in this article.
Full Annealing
Work hardening plays a major role in the shaping of metals. For example, in wire drawing process, while drawing metal wire through a die, the metal work hardens and the resistance to further deformation increases. The power required for continue drawing increases and finally the resistance to deformation equals the resistance to fracture and wire tends to break.
If drawing is stopped before drawing stress reaches fracture, it may be alright, but if it is necessary to further continue wire drawing either because the desired final size has not been obtained (final diameter is more than required diameter) or it is necessary to increase its electrical conductivity, it is required to return the material back to a condition approximating that it had prior to deformation.
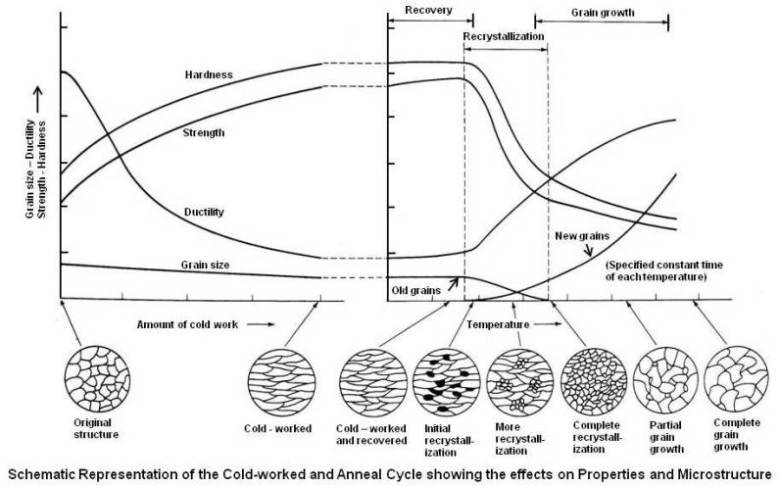
This is achieved by annealing. The process is carried out entirely in the solid state and is usually followed by slow cooling in the furnace from the desired temperature. The annealing process may be divided into three stages: recovery, recrystallization and grain growth.
Recovery
This is primarily a low temperature process, and the changes produced by it do not cause appreciable change in microstructure or the properties, such as tensile strength, yield strength, hardness and ductility. The principal effect of recovery is the relief of internal stresses due to cold working.
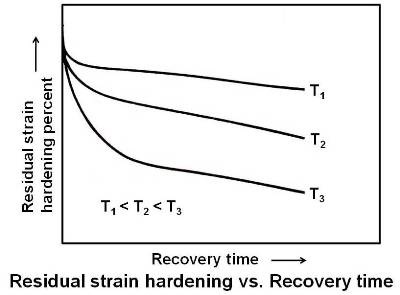
As shown in the above figure, at a given temperature, the rate of decrease in residual strain hardening is fastest at the beginning and drops off at longer times. Also, the amount of reduction in residual stress that occurs in a practical time increases with increasing temperature.
When the load which has caused plastic deformation in a polycrystalline material is released, all the elastic deformation does not disappear. This is due to the different orientation of the crystals, which will not allow some of them to move back when the load is released. As the temperature is increased, there is some spring back of these elastically displaced atoms which relieves most of the internal stress. In some cases there may be a slight amount of plastic flow, which may result in slight increase in hardness and strength. Electrical conductivity is also increased appreciably during the recovery stage.
Since the mechanical properties of the metal are essentially unchanged, the principal application of heating in the recovery range is in stress-relieving cold-worked alloys to prevent stress-corrosion cracking or to minimize the distortion produced by residual stresses in welded objects.
Commercially, this low temperature treatment in the recovery range is known as stress relief annealing. Recovery involves heating the objects to about 0.1 Tm, where Tm is the melting point of the metal in the absolute temperature scale.
Recrystallization
As the upper temperature of the recovery range is reached, the localized areas gain sufficient energy to overcome the rigidity of the distorted lattice and minute new crystals appear in the microstructure. These new crystals have the same composition and lattice structure as the original undeformed grains and are not elongated but are approximately uniform in dimensions (equiaxed). The new crystals generally appear at the most drastically deformed portions of the grain, usually the grain boundaries and slip planes. The cluster of atoms from which the new grains are formed is called a nucleus. Recrystallization takes place by a combination of nucleation of strain free grains and the growth of these nuclei to absorb the entire cold worked material.
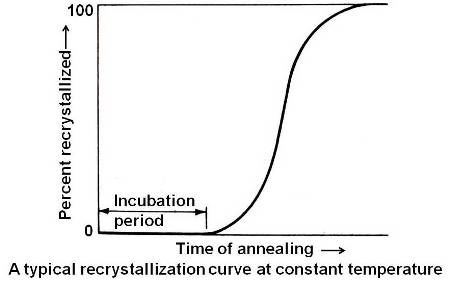
Above figure shows a typical recrystallization curve. This is a plot of the percent of the material recrystallized versus the time of annealing at constant temperature for a fixed composition and degree of cold working. This curve is typical of any process that occurs by nucleation and growth. Initially there is an incubation period in which enough energy is developed in order to start the process going. In this case, the incubation period is to allow the strain free nuclei to reach a visible microscopic size.
During recrystallization stage, there is a significant drop in tensile strength, hardness and a large increase in the ductility of the material.
Recrystallization Temperature
The term recrystallization temperature does not refer to a definite temperature below which recrystallization will not occur, but refers to the approximate temperature at which a highly cold worked material completely recrystallizes in one hour. The recrystallization temperatures of several metals and alloys are listed below.
| Material | Recrystallization Temperature (oF) |
|---|---|
| Copper (99.999 %) | 250 |
| Copper (5 % Zinc) | 600 |
| Aluminum (99.999 %) | 175 |
| Aluminum (99.0 % +) | 550 |
| Aluminum alloys | 600 |
| Iron (electrolytic) | 750 |
| Low carbon steel | 1000 |
| Zinc | 50 |
| Tin | 25 |
| Lead | 25 |
Pure metals have low recrystallization temperatures as compared with impure metals and alloys. Zinc, tin and lead have recrystallization temperatures below room temperature. This means that these metals cannot be cold worked at room temperature since they crystallize spontaneously, reforming a strain free lattice structure.
Recrystallization usually occurs at temperature of about 0.3 Tm in pure metals and of about 0.5 Tm in alloys where Tm is the melting temperature.
The greater the amount of prior deformation, the lower the temperature for the start of recrystallization, since there will be greater distortion and more internal energy left.
Increasing the annealing time decreases the recrystallization temperature for the start of recrystallization.
The recrystallization process is far more sensitive to change in temperature than to variation in time at constant temperature. For example, reduction in tensile strength of highly cold worked copper from 60000 psi (initial) to 40000 psi can be obtained by application of heat for 12 hours at 300° F, 6 hours at 320° F, 2 hours at 340° F, 1 hours at 370° F and 1/2 hour at 390° F. It may be noted that reduction in tensile strength is due to recrystallization.
Since for equal amounts of cold working, more strain hardening is introduced into initially fine gained metal than into initially coarse grained metal, the finer the grain size the lower the recrystallization temperature.
A certain minimum amount of cold working (usually 2 to 8 percent) is necessary for recrystallization. This is known as the critical deformation. At degrees of deformation smaller than this, the number of recrystallization nuclei becomes very small.
Grain Growth
In this stage the tensile strength and hardness continue to decrease but at a much less rate than the recrystallization stage. As the temperature increases the major change observed during this stage is the growth of the grain boundaries. The grain grows by coalescence (growing into each other) of some of the new formed grains as shown in figure given below.
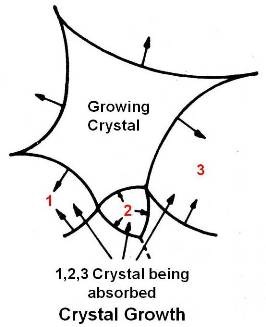
Growth does not start from nuclei but proceeds by gradual invasion of some grains by others. In other words, once the new equiaxed grains have been formed during recrystallization, if now the temperature is raised, some of these new grains will grow in size at the expense of their neighbouring grains. In grain growth, small grains grow still smaller and are ultimately absorbed by the large grains growing still larger. During grain growth the grain boundaries move towards their center of curvature (as against recrystallization where grain boundary movement is usually away from the center). The final gain size is more dependent on temperature than on time since grain growth normally continues (time permitting) to a grain size characteristic of the temperature.
Since annealing involves nucleation and grain growth, factors that favor rapid nucleation and slow growth will result in fine grained material and those which favor slow nucleation and rapid growth will result in coarse grain material. Grain growth largely depends on the following factors.
Degree of Prior Deformation
With increasing degree of deformation, an increasing number of points of high stress or high energy are present, leading to recrystallization from a large number of nuclei, and finally to a greater number of grains, and thus to a smaller grain size.
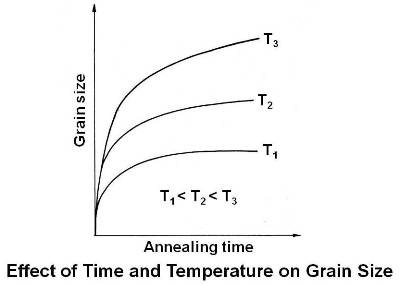
Time at Temperature and Annealing Temperature
Time at any temperature above the recrystallization temperature favors grain growth and increases the final grain size. The higher the temperature above the recrystallization temperature, the larger the grain size. These effects are shown in figure given below.
Heating Time
The shorter the time of heating to the annealing temperature, the finer the final grain size. Slow heating will form few nuclei, favoring grain growth and resulting in coarse grain.
Insoluble Impurities
The greater the amount and the finer the distribution of insoluble impurities the finer the grain size. They not only increase nucleation but act as barriers to the growth of grains.
The rate of cooling from the annealing temperature has a negligible effect on final grain size. The factor will be of interest only if the material has been heated far in the grain growth range and slow cooled. During slow cooling the material may have enough energy to continue grain growth, and some coarsening may result.
The production of coarse grains by growth after recrystallization is not generally desirable because formation of very large grains impairs mechanical properties.
Effect on Properties
Since full annealing resorts the material to a strain free lattice structure, it is essentially a softening process. Property changes produced by plastic deformation are removed, and the material returns very nearly to its original properties. Therefore, during annealing, the hardness and strength decreases, where as the ductility increases. The change in properties is shown schematically in the figure given below.
Hot Working
Hot working is usually described as working a material above its recrystallization temperature. The above definition, however does not take into account the rate of working.
When a material is plastically deformed, it tends to become harder, but the rate of hardening decreases as the temperature is increased. When a material is plastically deformed at an elevated temperature, two opposing effects take place at the same time – a hardening effect due to plastic deformation and a softening effect due to recrystallization. For a given rate of working, there must be some temperature at which these two effects will just balance. If the material is worked above this temperature, it is known as hot working, below this temperature it is known as cold working.
It may be noted that the terms hot and cold as applied to working do not have the same significance that they ordinarily have. For example, lead and tin, whose recrystallization temperature is below room temperature, may be hot worked at room temperature; but steel, with a high recrystallization temperature, may be cold worked at 1000° F.
Hot Working vs. Cold Working
Most of the metal shapes are produced from cast ingots. To manufacture sheet, plate, rod, wire, etc., from this ingot, the most economical method is by hot working. However, in the case of steel, the hot worked material reacts with oxygen as it cools down to room temperature and forms a characteristic dark oxide coating called scale. Occasionally, this scale may give difficulty during machining or forming operations. It is not possible to manufacture hot worked material to exact size because of dimensional changes that take place during cooling.
Cold worked material, on the other may be held to close tolerances. It is free of surface scales but requires more power for deformation and is therefore more expensive to produce.
Commercially, the initial reductions are carried out with the material at an elevated temperature, and the final reductions are done cold to take advantage of both processes.
