Information about three heat treatment operations on steel is given in this article. General information about annealing was given in the article titled annealing and hot working. In this article information about annealing as applicable to steel is given. Information about normalizing and hardening of steel is also given in this article. Information about other two heat treatment operations, tempering and case hardening are covered in a separate article.
Heat Treatment
The definition of heat treatment given in the Metals Handbook is: “A combination of heating and cooling operations timed and applied to a metal or alloy in the solid state in a way that will produce desired properties.†All basic heat-treating processes for steel involve the transformation or decomposition of austenite. The nature and appearance of these transformation products determine the physical and mechanical properties of any steel.
For heat treatment, the steel is first heated to some temperature above the critical temperature to form austenite. The rate of heating to desired temperature is not very important. However, highly stresses materials produced by cold work should be heated slowly than stress free material to avoid distortion. After heating, the material is cooled at a rate which will result in a desired operation (annealing, normalizing, hardening, etc.).
Full Annealing
This process consists of heating the steel to the proper temperature and then slowly cooling it through the transformation range preferably in the furnace or in any good heat-insulating material. The slow cooling is generally continued to low temperatures. The annealing is a very slow cooling process and therefore comes closest to following the iron-iron carbide equilibrium diagram. Annealing is carried out to refine grains, induce softness, improve electrical/magnetic properties and improve machinability.
A coarse-grained 0.20 percent carbon steel (hypoeutectoid steel) can be refined by full annealing as explained below.
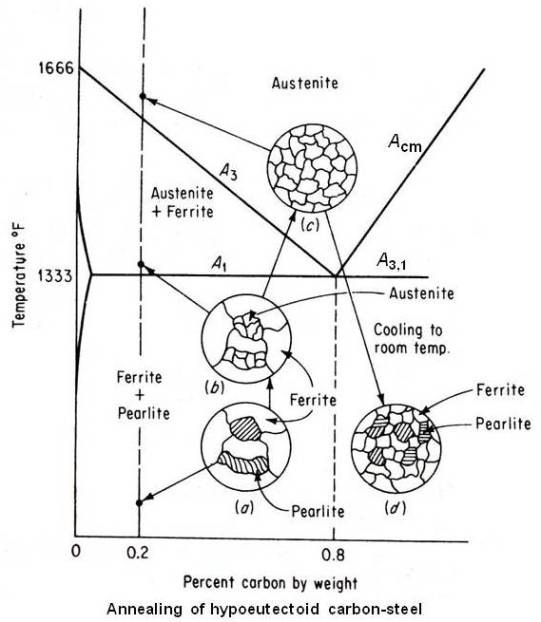
The microstructure of the coarse grain 0.2 percent carbon steel is shown at (a) in the above figure. When this steel is heated, no change will occur until it crosses lower critical temperature line A1. At that temperature, by eutectoid reaction pearlite areas will transform to small grains of austenite but the original large ferrite grains will remain unchanged as shown at (b) in above figure. To refine these large ferrite grains the material shall be further heated. Continued heating between the A1 and A3 lines will transform large ferrite grains to small grains of austenite so that above the A3 line (upper critical temperature line), the entire microstructure will show only small grains of austenite as shown at (c) in above figure. Now furnace cooling (very slow cooling) from this state will result in small grains of proeutectoid ferrite and small areas of coarse lamellar pearlite as shown at (d) in above figure. Therefore, the proper annealing temperature for full annealing of hypoeutectoid steels is approximately 50°F above the A3 line.
Refinement of the grain size of hypereutectoid steel will occur approximately about 50°F above the lower critical temperature (A3,1) line. Heating above this temperature will coarsen the austenitic grains, which, on cooling, will transform to large pearlite areas. The microstructure of annealed hypereutectoid steel will consist of coarse lamellar pearlite areas surrounded by a network of a proeutectoid cementite. Because this excess cementite network is brittle and tends to be plane of weakness, full annealing should never be a final heat treatment process for hypereutectoid steels. The presence of a thick and hard grain boundary will also result in poor machinability.
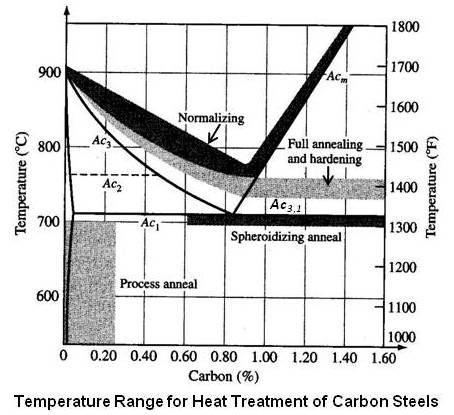
The proper full annealing temperature range for hypoeutectoid and hypereutectoid steels is shown in the figure given below. It also shows temperature range for other heat treatment operations – spheroidizing, process annealing, normalizing and hardening.
Spheroidizing
Annealed hypereutectoid steel with a microstructure of pearlite and cementite network generally gives poor machinability. Since cementite is hard and brittle, the cutting tool cannot cut through these plates. Instead, the plates have to be broken. Therefore, the tool is subjected to continual shock load by the cementite plates and results in a ragged surface finish. A heat-treating process which will improve the machinability is known as spheroidize annealing. This process will produce a spheroidal or globular form of carbide in a ferritic matrix as shown in the figure given below.
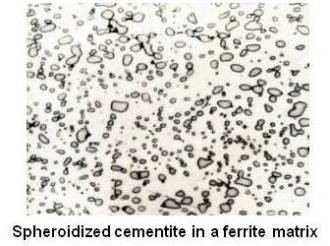
For spheroidizing, the hypereutectoid steel is held for a long time at a temperature just below the lower critical temperature line or is heated and cooled alternatively between temperatures that are just above and just below the lower critical line. Prolonged time at the elevated temperature will completely break up the pearlitic structure and cementite network. The cementite will become spheres. The cementite particles and the entire structure is called spheroidite. This structure is desirable when minimum hardness, maximum ductility and maximum machinability are required.
Low carbon steels are seldom spheroidized for machining, because they are excessively soft and gummy in the spheoridized conditions. The cutting tool will tend to push the material rather than cut it, causing excessive heat and wear on the cutting tip.
If steel is kept too long at spheroidize-annealing temperature, the cementite particles will coalesce and become elongated, thus reducing machinability.
Stress-relief Annealing
Stress-relief annealing is sometimes called subcritical annealing. It is useful in removing residual stresses due to heavy machining or other cold-working processes. It is usually carried out at temperatures below the lower critical temperature line. It is usually selected between 1000 and 1200°F.
Process Annealing
This heat treatment is used in the sheet and wire industries and is carried out by heating the steel to a temperature below the lower critical line (1000 to 1250°F). It is carried out after cold working to soften the steel by recrystallization for further working. It is very similar to stress-relief annealing.
Normalizing
The normalizing of steel is carried out by heating approximately 100°F above the upper critical temperature line (A3 or Acm) followed by cooling in still air to room temperature. The normalizing temperatures range is shown in the figure given earlier for annealing temperature. It may be noted that for hypereutectoid steels, it is necessary to heat it above the Acm line in order to dissolve the cementite network.
The purpose of normalizing is to produce harder and stronger steel than full annealing, so that for some applications normalizing may be a final heat treatment. Normalizing is also carried out to improve machinability.
The increase in cooling rate due to air cooling as compared to furnace cooling in case of full annealing affects the transformation of austenite and the resultant microstructure. Since the cooling is not carried out under equilibrium conditions, the iron-iron carbide diagram cannot be used to predict the proportions of proeutectoid ferrite and pearlite (in case of hypoeutectoid steel) or proeutectoid cementite and pearlite (in case of hypereutectoid steel) that will exist at room temperature.
There is less time for the formation of the proeutectoid constituent; consequently there will be less proeutectoid ferrite in normalized hypoeutectoid steels and less proeutectoid cementite in hypereutectoid steels as compared with annealed ones. For hypereutectoid steels, normalizing will reduce the continuity of the proeutectoid cementite network, and in some cases it may be suppressed entirely. Since it was the presence of the cementite network which reduced the strength of annealed hypereutectoid steels, normalized steels should show an increase in strength.
Apart from influencing the amount of proeutectoid constituents that will form, the faster cooling rate in normalizing will also affect the temperature of austenite transformation and the fineness of the pearlite. In general, the faster the cooling rate, the lower the temperature of austenite transformation and the finer the pearlite.
The difference in spacing of the cementite plates in the pearlite between annealing and normalizing is shown schematically in the figure given below.
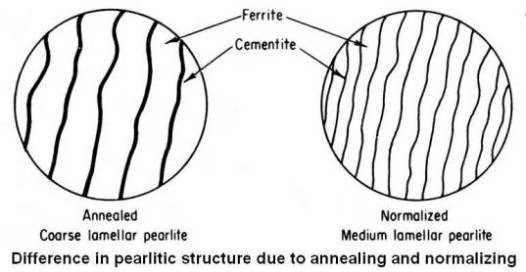
Ferrite is very soft, while cementite is very hard. With the cementite plates closer together in the case of normalized medium pearlite, they tend to stiffen the ferrite so it will not yield as easily, thus increasing hardness. If the annealed coarse pearlite has a hardness of about Rockwell C 10, then the normalized medium pearlite will have hardness of about Rockwell C 20.
Hardening
In this process, austenite is transformed to martensite as a result of quenching (rapid cooling) from furnace to room temperature.
The recommended austenitizing temperature for hypoeutectoid steels is about 50°F above the upper critical temperature line. This is the same as the recommended annealing temperature. For plain-carbon hypereutectoid steel the recommended austenitizing temperature is usually between the Acm and A3,1 lines (about 50°F above the A3,1 line, same as the recommended annealing temperature); therefore, undissolved carbides would tend to be present in the microstructure at room temperature. The Acm line rises so steeply that an excessively high temperature may be required to dissolve all the proeutectoid cementite in the austenite. This tends to develop undesirable coarse austenite grain size, with danger of cracking on cooling.
Under slow or moderate cooling rates, the carbon atoms are able to diffuse out of the austenite structure. The iron atoms then move slightly to become b.c.c. (body-centered cubic). This gamma to alpha transformation takes place by a process of nucleation and growth and is time dependent. With a still further increase in cooling rate, insufficient time is allowed for the carbon to diffuse out of solution, and although some movement of iron atoms takes place, the structure cannot become b.c.c. while the carbon is trapped in solution. The resultant structure, called martensite is supersaturated solid solution of carbon trapped in a less dense (than b.c.c.) body-centered tetragonal structure. In this structure, two dimensions of the unit cell are equal, but the third is slightly extended because of the trapped carbon. This highly distorted lattice structure is the prime reason for the high hardness of martensite. After drastic cooling (quenching), martensite appears microscopically as a white needlelike or acicular structure sometimes described as a pile of straw. In most steels, the martensite structure appears vague and unresolvable. A typical microstructure of martensite is shown in the figure given below.

In quenching at low temperature, the molecular mobility, i.e. diffusion, becomes too small for the formation of nuclei. Therefore, the austenite changes into a distorted body-centered structure, with little or no diffusion of the carbon into particles of cementite, to form martensite. The plates of martensite are formed at a high speed (less than 0.002 sec). This suggests that the mechanism of formation of this structure is not nucleation and growth but a shearing process. This resembles the process of mechanical twinning and involves very little atomic movement, but considerable internal stress due to the shear and to the position of the carbon atoms. As the temperature decreases the elastic energy increases and eventually causes a shear in a part of the matrix, which stabilizes the rest. Further shear can only occur when the temperature is lowered and more energy gained. The amount of martensite formed, therefore, is practically independent of time and depends principally on the temperatures at which the steel is held.
Hence a proportion of austenite is usually retained in quenched steel which can be reduced in amount by a decrease in temperature. This fact is used in sub-zero quenching.
Several important characteristics of martensite transformation are as under.
The transformation is diffusionless, and there is no change in chemical composition. Small volumes of austenite suddenly change crystal structure by shearing action.
The transformation proceeds only during cooling and ceases if cooling is interrupted. Therefore, the transformation depends only upon the decrease in temperature and is independent of time. A transformation of this type is said to be athermal, in contrast to one that will occur at constant temperature (isothermal transformation). The amount of martensite formed with decreasing temperature is not linear. The number martensite needles produced at first is small, then the number increases, and finally, near the end, it decreases again. The temperature of the start of martensite formation is known as Ms temperature and that of the end of martensite formation is known as the Mf temperature. If the steel is held at any temperature below the Ms, the transformation to martensite will not proceed again unless the temperature is dropped.
The martensite transformation of a given alloy cannot be suppressed, nor can the Ms, temperature be changed by changing the cooling rate. The temperature range of the formation of martensite is characteristic of a given alloy. The Ms temperature seems to be a function of chemical composition only. There are several formulas. One such formula is as under.
Ms (°F) = 1000 – (650 x %C) – (70 x %Mn) – (35 x %Ni) – (70 x %Cr) – (50 x %Mo)
Martensite is probably never in a condition of real equilibrium, although it may persist indefinitely at or near room temperature. The structure can be considered as a transition between the unstable austenite phase and the final equilibrium condition of a mixture of ferrite and cementite.
Although martensite is always harder than the austenite from which it forms, extreme hardnesses are possible only in steels that contain sufficient carbon. As shown in the figure given below, the hardness of martensite increases rapidly at first with increase in carbon content, reaching about Rockwell C 60 at 0.4 percent carbon. Beyond that point the curve levels off, and at the eutectoid composition, it is about Rockwell C 65.
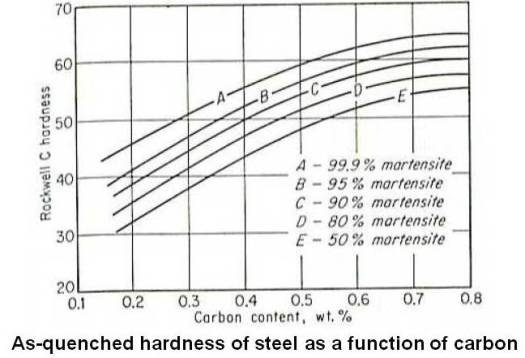
The high hardness of martensite is believed to be a result of the severe lattice distortions produced by its formation, since the amount of carbon present is many times more than can be held in solid solution.
Tempering
Steels can be heat treated to high hardness and strength levels. Hardening is carried out because structural components subjected to high operating stress need the high strength of a hardened structure. Similarly, tools such as dies, knives, cutting devices, and forming devices need a hardened structure to resist wear and deformation.
However, the as-quenched hardened steels are too brittle for most applications. Tempering is a heat treatment process that reduces the brittleness of hardened steel without significantly lowering its hardness and strength. In view of this, all hardened steels are tempered before use. More information about tempering is covered in a separate article on it.
